Abstract
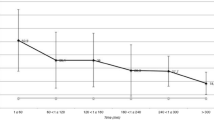
The aim of the present study was to quantify in man the distribution and clearance of two aqueous sodium hyaluronate (SH) solutions of 0.125% and 0.250% after the administration of 25 μl onto the cornea. Isotonic phosphate buffer (PB) was used as a reference instillation. No systemic or local medication was given to the seven 18- to 30-year-old, healthy male volunteers. A detailed evaluation of the anterior segment of the eye, as well as a Schirmer test and a break-up time measurement, yielded results within the normal range. The clearance of 0.125% and 0.250% SH solutions radiolabelled with sodium pertechnetate Tc-99m was measured by gamma scintigraphy and compared with that of a PB solution tagged with the same radiolabel. There was no statistically significant difference between the quantities of 0.125% SH and PB solutions remaining in the precorneal space at 20 min (paired t-test,P=0.78,n=7). However, in comparing the 0.250% SH with the PB solution, we observed a statistically significant difference (P=0.01,n=7) in the amount remaining in the precorneal space after the same interval. Actually, 53% of the radiolabelled 0.250% SH solution remained on the cornea as compared with 30% for the 0.125% SH solution and 18.3% for the PB solution. These results suggest that an SH solution of 0.250% might have a prolonged residence time on the precorneal surface, and that SH could therefore be used as an additive in various drug-release systems for the eye.
Similar content being viewed by others
References
Barett TW (1976) The molecular dynamics of hyaluronates in solution. Biosystems 8:103–109
Camber O, Lundgren P (1985) Diffusion of some low molecular weight compounds in sodium hyaluronate. Acta Pharm Suec 22:315–320
Camber O, Edman P, Gurny R (1987) Influence of sodium hyaluronate on the miotic effect of pilocarpine in rabbits. Curr Eye Res 6:779–783
Gurny R, Ibrahim H, Aebi A, Buri P, Wilson CG, Washington N, Edman P, Camber O (1987) Design and evaluation of controlled release systems for the eye. J Controlled Release 6:367–373
Norn SM (1981) Perioperative protection of cornea and conjunctiva. Acta Ophthalmol 59:587–594
Polack MF, McNiece MT (1982) The treatment of dry eyes with Na hyaluronate (Healon®). Cornea 1:133–136
Robinson JR (1989) Ocular drug delivery mechanism(s) of corneal drug transport and mucoadhesive delivery systems. STP Pharma 5:839–846
Rozier A, Mazuel C, Grove J, Plazonnet B (1989) Gelvrite: a novel, ion-actived, in-situ gelling polymer for ophthalmic vehicoles. Effect on bioavailability of timolol. Int J Pharm 57:163–168
Saettone MF, Giannaccini B, Delmonte G, Campigli V, Tota G, La Marca F (1988) Solubilization of tropicamide by poloxamers: physicochemical data and activity data in rabbits and humans. Int J Pharm 43:67–76
Sand BB, Marner K, Norn MS (1989) Sodium hyaluronate in the treatment of kerato-conjunctivitis sicca. Acta Ophthalmol 67:181–183
Saric D, Reim M (1984) Behandlung von Verätzungen des vorderen Augenabschnitts mit hochpolymeren Na-Hyaluronat (Healon®). Fortschr Ophthalmol 81:588–591
Tabatabay C (1984) Utilisation du Healon® lors d'éépithélialisation cornéenne défectueuse. J Fr Ophtalmol 7:755
Tabatabay C (1985) Instillation d'acide hyaluronique à 0.1% lors de kératite sèche sévère. J Fr Ophthalmol 8:513
Tabatabay C, Ryser JE, Gurny R, Edman P, Camber O (1989) Precorneal residence time of sodium hyaluronate solutions measured by gamma scintigraphy. Invest Ophthalmol Vis Sci [Suppl] 30:248
Wysenbeek YS, Loya N, Ben Sira I, Ophir I, Ben Shaul Y (1988) The effect of sodium hyaluronate on the cornea epithelium. Invest Ophthalmol Vis Sci 29:194–199
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gurny, R., Ryser, J.E., Tabatabay, C. et al. Precorneal residence time in humans of sodium hyaluronate as measured by gamma scintigraphy. Graefe's Arch Clin Exp Ophthalmol 228, 510–512 (1990). https://doi.org/10.1007/BF00918481
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00918481