Abstract
Background/Purpose
To evaluate the effect of individualized repeated intravitreal injections of ranibizumab (Lucentis) on visual acuity (VA) and central foveal thickness (CFT) for central retinal vein occlusion (CRVO)-induced macular edema.
Methods
Our study was a prospective interventional case series. Twelve eyes of 12 consecutive patients diagnosed with CRVO-related macular edema (nine perfused, three ischemic CRVO) treated with repeated (when CFT was >220 μm) intravitreal injections of ranibizumab as a monotherapy within 3 months of onset were evaluated. Optical coherence tomography (OCT) and fluorescein angiography (FA) were performed monthly and every 3 months respectively. Changes in VA (ETDRS) and CFT were analyzed using the student’s paired t-test.
Results
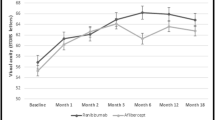
The mean time from diagnosis until injection was 80 days (2.7 months; range, 63–90 days) and the follow-up time was 12 months. In total, 89 injections were performed (mean 7.4). The mean CFT improved from 480 ± 166 μm at baseline to 230 ± 33 μm (P < 0.001) at the end of the follow-up. During the same period, of the 12 eyes, eight demonstrated improved VA (>0.3 LogMAR change, >15 letters), three stable VA and one worse VA as compared to baseline. None of the nine patients with perfused CRVO were converted to ischemic at 12 months, and one of the three eyes with ischemic CRVO developed iris neovascularization despite two ranibizumab injections. No ocular or systemic side-effects were noted.
Conclusion
Individualized repeated intravitreal injections of ranibizumab have shown promising results in VA improvement and decrease in CFT in patients with macular edema associated with CRVO. Further studies are needed in order to elucidate the role of intravitreal lucentis in the ischemic form of CRVO, and its efficacy in preventing conversion from the perfused to the ischemic form of the disease.




Similar content being viewed by others
References
The Central Vein Occlusion Study Group (1995) A randomized clinical trial of early panretinal photocoagulation for ischemic central vein occlusion. The Central Vein Occlusion Study Group N report. Ophthalmology 102:1434–1444
The Central Vein Occlusion Study Group (1995) Evaluation of grid pattern photocoagulation for macular edema in central vein occlusion. The Central Vein Occlusion Study Group M report. Ophthalmology 102:1425–1433
Jonas JB (2005) Intravitreal triamcinolone acetonide for treatment of intraocular oedematous and neovascular diseases. Acta Ophthalmol Scand 83:645–663
Jonas JB, Akkoyun I, Kamppeter B, Kreissig I, Degenring RF (2005) Intravitreal triamcinolone acetonide for treatment of central retinal vein occlusion. Eur J Ophthalmol 15:751–758
Gregori NZ, Rosenfeld PJ, Puliafito CA, Flynn HW Jr, Lee JE, Mavrofrides EC, Smiddy WE, Murray TG, Berrocal AM, Scott IU, Gregori G (2006) One-year safety and efficacy of intravitreal triamcinolone acetonide for the management of macular edema secondary to central retinal vein occlusion. Retina 26:889–895
Goff MJ, Jumper JM, Yang SS, Fu AD, Johnson RN, McDonald HR, Ai E (2006) Intravitreal triamcinolone acetonide treatment of macular edema associated with central retinal vein occlusion. Retina 26:896–901
Patel PJ, Zaheer I, Karia N (2008) Intravitreal triamcinolone acetonide for macular oedema owing to retinal vein occlusion. Eye 22:60–64
Ramezani A, Entezari M, Moradian S, Tabatabaei H, Kadkhodaei S (2006) Intravitreal triamcinolone for acute central retinal vein occlusion; a randomized clinical trial. Graefes Arch Clin Exp Ophthalmol 24:1601–1606
Moschos MM, Brouzas D, Loukianou E, Apostolopoulos M, Moschos M (2007) Intraocular triamcinolone acetonide for macular edema due to CRVO. A multifocal-ERG and OCT study. Doc Ophthalmol 114:1–7
Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE et al (1994) Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Eng J Med 331:1480–1487
Frank RN, Amin RH, Eliott D, Puklin JE, Abrams GW (1996) Basic fibroblast growth factor and vascular endothelial growth factor are present in epiretinal and choroidal neovascular membranes. Am J Ophrhalmol 122:393–403
Keck PJ, Hauser SD, Krivi G, Sanzo K, Warren T, Feder J, Connolly DT (1989) Vascular permeability factor, an endothelial cell mitogen related to PDGF. Science 246:1309–1312
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N (1989) Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246:1306–1309
Senger DR, Connolly DT, Van de Water L, Feder J, Dvorak HF (1990) Purification and NH2-terminal amino acid sequence of guinea pig tumor-secreted vascular permeability factor. Cancer Res 50:1774–1778
Nguyen QD, Tatlipinar S, Shah SM, Haller JA, Quinlan E, Sung J, Zimmer-Galler I, Do DV, Campochiaro PA (2006) Vascular endothelial growth factor is a critical stimulus for diabetic macular edema. Am J Ophthalmol 142:961–969
Iturralde D, Spaide RF, Meyerle CB, Klancnik JM, Yannuzzi LA, Fisher YL, Sorenson J, Slakter JS, Freund KB, Cooney M, Fine HF (2006) Intravitreal bevacizumab (Avastin) treatment of macular edema in central retinal vein occlusion: a short-term study. Retina 26:279–284
Jaissle GB, Ziemssen F, Petermeier K, Szurman P, Ladewig M, Gelisken F, Völker M, Holz FG, Bartz-Schmidt KU (2006) Bevacizumab for treatment of macular edema secondary to retinal vein occlusion. Ophthalmologe 103:471–475
Stahl A, Agostini H, Hansen LL, Feltgen N (2007) Bevacizumab in retinal vein occlusion-results of a prospective case series. Graefes Arch Clin Exp Ophthalmol 245:1429–1436
Priglinger SG, Wolf AH, Kreutzer TC, Kook D, Hofer A, Strauss RW, Alge CS, Kunze C, Haritoglou C, Kampik A (2007) Intravitreal bevacizumab injections for treatment of central retinal vein occlusion: six-month results of a prospective trial. Retina 27:1004–1012
Ferrara DC, Koizumi H, Spaide RF (2007) Early bevacizumab treatment of central retinal vein occlusion. Am J Ophthalmol 14:864–871
Hsu J, Kaiser RS, Sivalingam A, Abraham P, Fineman MS, Samuel MA, Vander JF, Regillo CD, Ho AC (2007) Intravitreal bevacizumab (avastin) in central retinal vein occlusion. Retina 27:1013–1019
Pieramici DJ, Rabena M, Castellarin AA, Nasir M, See R, Norton T, Sanchez A, Risard S, Avery RL (2008) Ranibizumab for the treatment of macular edema associated with perfused central retinal vein occlusions. Ophthalmology 115:e47–e54
Campochiaro PA, Hafiz G, Shah SM, Nguyen QD, Ying H, Do DV, Quinlan E, Zimmer-Galler I, Haller JA, Solomon SD, Sung JU, Hadi Y, Janjua KA, Jawed N, Choy DF, Arron JR (2008) Ranibizumab for macular edema due to retinal vein occlusions: implication of VEGF as a critical stimulator. Mol Ther 16:791–799
Spaide RF, Chang LK, Klancnik JM, Yannuzzi LA, Sorenson J, Slakter JS, Freund KB, Klein R (2008). Prospective study of intravitreal ranibizumab as a treatment for decreased visual acuity secondary to central retinal vein occlusion. Am J Ophthalmol 147:298–306
Brown DM, Kaiser PK, Michels M, Soubrane G, Heier JS, Kim RY, Sy JP, Schneider S, ANCHOR Study Group (2006) Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 355:1432–1444
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY, MARINA Study Group (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355:1419–1431
El-Ashry M, Hegde V, James P, Pagliarini S (2008) Analysis of macular thickness in British population using optical coherence tomography (OCT): an emphasis on interocular symmetry. Curr Eye Res 33(8):693–699
Kim BY, Smith SD, Kaiser PK (2006) Optical coherence tomographic patterns of diabetic macular edema. Am J Ophthalmol 142:405–412
The Central Vein Occlusion Study Group (1997) Natural history and clinical management of central retinal vein occlusion. The Central Vein Occlusion Study Group. Arch Ophthalmol 115:486–491
Food and Drug Administration (2006). www.fda.gov/cder/foi/label/2006/125156lbl.pdf
The Central Vein Occlusion Study Group (1993) Baseline and early natural history report. The Central Vein Occlusion Study. Arch Ophthalmol. 111:1087–1095
Author information
Authors and Affiliations
Corresponding author
Additional information
No financial or proprietary interest by any of the authors
Rights and permissions
About this article
Cite this article
Rouvas, A., Petrou, P., Vergados, I. et al. Intravitreal ranibizumab (Lucentis) for treatment of central retinal vein occlusion: a prospective study. Graefes Arch Clin Exp Ophthalmol 247, 1609–1616 (2009). https://doi.org/10.1007/s00417-009-1138-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-009-1138-y