Summary
Background
To assess whether the period between initial symptoms and therapy with ranibizumab in patients with choroidal neovascularization (CNV) influences visual outcome after a follow-up of 12 months.
Methods
Fifty patients with CNV were retrospectively split into three groups depending on the duration of visual symptoms: group I: < 1 month, group II: 1–6 months, and group III: > 6 months. Best-corrected visual acuity (BCVA) and central retinal thickness (CRT) were recorded at baseline, 2, 6, and 12 months. Patients received two initial intravitreal injections of 0.5 mg ranibizumab at baseline and reinjections as needed.
Results
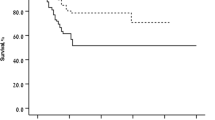
The mean time span between initial symptoms and treatment was 66 ± 63 days. A longer duration of visual symptoms was significantly correlated with a lower BCVA at baseline, but also after 6 and 12 months.
Conclusions
Shorter duration of visual symptoms prior to treatment is associated with a better visual outcome.
Zusammenfassung
Hintergrund
Ziel dieser Studie war es, einen möglichen zeitlichen Einfluss zwischen Auftreten von Symptomen und Beginn einer Therapie bei choroidaler Neovaskularisation (CNV) auf das Sehvermögen nach einem Beobbachtungszeitraum von 12 Monaten herauszufinden.
Methodik
Fünfzig PatientInnen mit CNV wurden retrospektiv in drei Gruppen abhängig von der Symptomdauer geteilt: Gruppe I: < 1 Monat, Gruppe II: 1–6- Monate, Gruppe III: > 6 Monate. Der bestkorrigierte Visus und die zentrale Netzhautdicke wurden zu Beginn, nach 2, 6 und 12 Monaten erhoben. Die PatientInnen erhielten zwei initiale Injektionen mit 0,5 mg Ranibizumab in den Glaskörper. Weitere Injektionen wurden bei Bedarf verabreicht.
Ergebnisse
Die durchschnittliche Dauer zwischen Erstauftreten der Symptome und Beginn der Behandlung betrug 66 ± 63 Tage. Eine längere Symptomdauer korrelierte signifikant mit einem schlechteren Ausgangsvisus, aber auch mit einem schlechteren Sehvermögen nach 6 und 12 Monaten.
Schlussfolgerungen
Je kürzer die Symptomdauer vor Beginn der Behandlung ist, desto besser ist das Visusergebnis nach 12 Monaten.

Similar content being viewed by others
References
Kahn HA, Leibowitz HM, Ganley JP, et al. The Framingham Eye Study. I. Outline and major prevalence findings. Am J Epidemiol. 1977;106:17–32.
Klaver CC, Wolfs RC, Vingerling JR, Hofman A, de Jong PT. Age-specific prevalence and causes of blindness and visual impairment in an older population: the Rotterdam Study. Arch Ophthalmol. 1998;116:653–8.
Spitzer MS, Ziemssen F, Bartz-Schmidt KU, Gelisken F, Szurman P. Treatment of age-related macular degeneration: focus on ranibizumab. Clin Ophthalmol. 2008;2:1–14.
Macular Photocoagulation Study Group. Five-year follow-up of fellow eyes of patients with age-related macular degeneration and unilateral extrafoveal choroidal neovascularization. Arch Ophthalmol. 1993;111:1189–99.
Barouch FC, Miller JW. Anti-vascular endothelial growth factor strategies for the treatment of choroidal neovascularization from age-related macular degeneration. Int Ophthalmol Clin. 2004;44:23–32.
Rosenfeld PJ, Brown DM, Heier JS, et al. MARINA Study Group. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355:1419–31.
Furino C, Boscia F, Recchimurzo N, et al. Intravitreal bevacizumab for treatment-naive subfoveal occult choroidal neovascularization in age-related macular degeneration. Acta Ophthalmol. 2009;87:404–7.
Kaiser PK. Antivascular endothelial growth factor agents and their development: therapeutic implications in ocular diseases. Am J Ophthalmol. 2006;42:660–8.
Ladewig MS, Karl SE, Hamelmann V, et al. Combined intravitreal bevacizumab and photodynamic therapy for neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2008;246:17–25.
Ng EW, Adamis AP. Targeting angiogenesis, the underlying disorder in neovascular age-related macular degeneration. Can J Ophthalmol. 2005;40:352–68.
Singer MA, Awh CC, Sadda S, et al. HORIZON: An open-label extension trial of ranibizumab for choroidal neovascularization secondary to age-related macular degeneration. Ophthalmology. 2012;119(6):1175–83.
Brown DM, Kaiser PK, Michels M, et al. ANCHOR Study Group. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med. 2006;355:1432–44.
Boyer DS, Antoszyk AN, Awh CC, Bhisitkul RB, Shapiro H, Acharya NR. MARINA Study Group. Subgroup analysis of the MARINA study of ranibizumab in neovascular age-related macular degeneration. Ophthalmology. 2007;114:246–52.
Rosenfeld PJ, Rich RM, Lalwani GA. Ranibizumab: Phase III clinical trial results. Ophthalmol Clin North Am. 2006;19:361–72.
Lalwani GA, Rosenfeld PJ, Fung AE, et al. A variable-dosing regimen with intravitreal ranibizumab for neovascular age-related macular degeneration: year 2 of the PrONTO Study. Am J Ophthalmol. 2009;148(1):43–58.
Klein ML, Jorizzo PA, Watzke RC. Growth features of choroidal neovascular membranes in age-related macular degeneration. Ophthalmology. 1989;96:1416–9.
Vander JF, Morgan CM, Schatz H. Growth rate of subretinal neovascularization in age-related macular degeneration. Ophthalmology. 1989;96:1422–6.
Muether PS, Hermann MM, Koch K, Fauser S. Delay between medical indication to anti-VEGF treatment in age-related macular degeneration can result in a loss of visual acuity. Graefes Arch Clin Exp Ophthalmol. 2011;249:633–7.
Schalnus R, Meyer CH, Kuhli-Hattenbach C, Luchtenberg M. Time between symptom onset and assessment in age-related macular degeneration with subfoveal choroidal neovascularization. Ophthalmologica. 2010;224:176–82.
Oliver-Fernandez A, Bakal J, Segal S, Shah GK, Dugar A, Sharma S. Progression of visual loss and time between initial assessment and treatment of wet age-related macular degeneration. Can J Ophthalmol. 2005;40:313–9.
Algvere PV, Steén B, Seregard S, Kvanta A. A prospective study on intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration of different durations. Acta Ophthalmol. 2008;86:482–9.
Rauch R, Weingessel B, Maca SM, Vécsei-Marlovits PV. Time to first treatment—the significance of early treatment of exudative age related macular degeneration. Retina. 2011;32(7):1260–4.
Heimes B, Lommatzsch A, Zeimer M, et al. Long-term visual course after anti-VEGF therapy for exudative AMD in clinical practice evaluation of the German reinjection scheme. Graefes Arch Clin Exp Ophthalmol. 2011;249:639–44.
Dadgostar H, Ventura AA, Chung JY, Sharma S, Kaiser PK. Evaluation of injection frequency and visual acuity outcomes for ranibizumab monotherapy in exudative age-related macular degeneration. Ophthalmology. 2009;116:1740–7.
Conflict of interest
The authors have no proprietary interest in any material presented in the study. Pia Veronika Vecsei-Marlovits and Birgit Weingessel received support by the Novartis Company for presentations and consultancy not associated with this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weingessel, B., Hintermayer, G., Maca, S. et al. The significance of early treatment of exudative age-related macular degeneration: 12 months’ results. Wien Klin Wochenschr 124, 750–755 (2012). https://doi.org/10.1007/s00508-012-0249-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-012-0249-3
Keywords
- choroidal neovascularization
- age-related macular degeneration
- as-needed
- duration of symptoms
- ranibizumab
- time to first treatment