Abstract
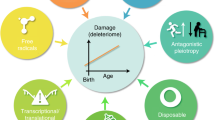
Recent experimental work from a variety of biological systems, ranging from yeast to human beings, lends increasing support to the view that stochastic damage inflicted to biological macromolecules is the driving force for the ageing process. The damage is derived from small reactive molecules, most prominently reactive oxygen intermediates (ROD, that arise during normal cellular metabolism and are associated with important if not essential cellular functions. The major classes of macromolecules at risk are proteins, lipids and DNA, but damage to DNA (both nuclear and mitochondrial) may entail particularly severe consequences. Cellular dysfunction resulting from macromolecular damage can be detected as a variety of expressions, such as genomic instability, inappropriate cell differentiation events or cell death. While for post-mitotic cell types replacement of the dead cell by another cell of the same lineage is not possible, mitotic cell types may initially replace dead cells via cell proliferation. But exhaustion of the self-renewal capacity of the respective lineage, by either replication-associated or damage-associated telomere shortening, will ultimately also lead to loss of parenchymal cell mass and functional impairment of tissues, the latter being a typical feature of ageing of tissues and organs. It has been demonstrated in various experimental systems that the rate ageing of can be retarded by lowering the production of endogenous ROI or by improving cellular anti-oxidative defences. Whether augmentation of cellular DNA repair capacity will have the same effect remains to be seen.
Similar content being viewed by others
Article PDF
References
Bürkle A . Maintaining the stability of the genome. In: Rattan SIS, Toussaint O, editors. Molecular gerontology: Research status and strategies. New York: Plenum Press, 1996:25–36.
Jazwinski SM . Molecular mechanisms of yeast longevity. Trends Microbiol 1999;7:247–52.
Guarente L . Sir2 links chromatin silencing, metabolism, and aging. Genes Dev 2000;14:1021–6.
Osiewacz HD, Borghouts C . Mitochondrial oxidative stress and aging in the filamentous fungus Podospora anserina. Ann N Y Acad Sci 2000;908:31–9.
Murakami S, Tedesco PM, Cypser JR, Johnson TE . Molecular genetic mechanisms of life span manipulation in Caenorhabditis elegans. Ann N Y Acad Sci 2000;908:40–9.
Lakowski B, Hekimi S . The genetics of caloric restriction in Caenorhabditis elegans. Proc Natl Acad Sci USA 1998;95:13091–6.
Melov S, Ravenscroft J, Malik S, Gill MS, Walker DW, Clayton PE, et al. Extension of life-span with Superoxide dismutase/catalase mimetics. Science 2000;289:1567–9.
Orr WC, Sohal RS . Extension of life-span by overexpression of Superoxide dismutase and catalase in Drosophila melanogaster. Science 1994;263:1128–30.
Brack C, Bechter-Thuring E, Labuhn M . N-Acetylcysteine slows down ageing and increases the life span of Drosophila melanogaster. Cell Mol Life Sci 1997;53:960–6.
Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, Utsugi T, et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 1997;390:45–50.
Vijg J, Dolle ME, Martus HJ, Boerrigter ME . Transgenic mouse models for studying mutations in vivo: applications in aging research. Mech Ageing Dev 1997;99:257–71.
Rudolph KL, Chang S, Lee HW, Blasco M, Gottlieb GJ, Greider C, et al. Longevity, stress response and cancer in ageing telomerase-deficient mice. Cell 1999;96:701–12.
Migliaccio E, Giorgio M, Mele S, Pelicci G, Reboldi P, Pandolfi PP, et al. The p66shc adaptor protein controls oxidative stress response and life span on mammals. Nature 1999;402:309–13.
Martin K, Potten CS, Kirkwood TB . Age-related changes in irradiation-induced apoptosis and expression of p21 and p53 in crypt stem cells of murine intestine. Ann N Y Acad Sci 2000;908:315–8.
Sohal RS, Weindruch R . Oxidative stress, caloric restriction, and aging. Science 1996;273:59–63.
Merry BJ . Calorie restriction and age-related oxidative stress. Ann N Y Acad Sci 2000;908:180–98.
Gonos ES, Derventzi A, Kveiborg M, Agiostratidou G, Kassem M, Clark BF, et al. Cloning and identification of genes that associate with mammalian replicative senescence. Exp Cell Res 1998;240:66–74.
Lane MA, Ingram DK, Roth GS . Nutritional modulation of aging in nonhuman primates. J Nutr Health Aging 1999;3:69–76.
Yu CE, Oshima J, Fu YH, Wijsman EM, Hisama F, Alisch R, et al. Positional cloning of the Werner's syndrome gene. Science 1996;272:258–62.
Nehlin JO, Skovgaard GL, Bohr VA . The Werner syndrome: a model for the study of human aging. Ann N Y Acad Sci 2000;908:167–79.
Mukherjee AB, Costello C . Aneuploidy analysis in fibroblasts of human premature aging syndromes by FISH during in vitro cellular aging. Mech Ageing Dev 1998;103:209–22.
de Boer J, Hoeijmakers JH . Cancer from the outside, aging from the inside: mouse models to study the consequences of defective nucleotide excision repair. Biochimie 1999;81:127–37.
Yashin AI, De Benedicts G, Vaupel JW, Tan Q, Andreev KF, Iachine IA, et al. Genes and longevity: lessons from studies of centenarians. J Gerontol A Biol Sci Med Sci 2000;55:B319–28.
Muiras ML, Müller M, Schächter F, Bürkle A . Increased poly(ADP-ribose) polymerase activity in lymphoblastoid cell lines from centenarians. J Mol Med 1998;76:346–54.
Hayflick L . The cell biology of aging. J Invest Dermatol 1979;73:8–14.
Harley CB, Futcher AB, Greider CW . Telomeres shorten during ageing of human fibroblasts. Nature 1990;345:458–60.
Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB, et al. Extension of life-span by introduction of telomerase into normal human cells. Science 1998;279:349–52.
Campisi J . Cancer, ageing and cellular senescence. In Vivo 2000;14:183–8.
von Zglinicki T . Role of oxidative stress in telomere length regulation and replicative senescence. Ann N Y Acad Sci 2000;908:99–110.
Kletsas D, Pratsinis H, Zervolea I, Handris P, Sevaslidou E, Ottaviani E, et al. D. Fibroblast responses to exogenous and autocrine growth factors relevant to tissue repair: the effect of aging. Ann N Y Acad Sci 2000;908:155–66.
Toussaint O, Dumont P, Dierick JF, Pascal T, Frippiat C, Chainiaux F, et al. Stress-reduced premature senescence: essence of life, evolution, stress, and aging. Ann N Y Acad Sci 2000;98:85–98.
Atamna H, Paler-Martinez A, Ames BN . N-t-Butyl hydroxylamine, a hydrolysis product of alpha-phenyl-N-t-butyl nitrone, is more potent in delaying senescence in human lung fibroblasts. J Biol Chem 2000;275:6741–8.
Martin GM, Austad SN, Johnson TE . Genetic analysis of ageing: role of oxidative damage and environmental stresses. Nature Genet 1996;13:25–34.
Shigenaga MK, Hagen TM, Ames BN . Oxidative damage and mitochondrial decay in aging. Proc Natl Acad Sci USA 1994;91:10771–8.
Kopsidas G, Kovalenko SA, Heffernan DR, Yarovaya N, Kramarova L, Stojanovski D, et al. Tissue mitochondrial DNA changes: a stochastic system. Ann N Y Acad Sci 2000;908:226–43.
Riggs JE . Carcinogenesis, genetic instability and genomic entrophy: insight derived from malignant brain tumor age specific mortality rate dynamics. J Theor Biol 1994;170:331–8.
Ku HH, Brunk UT, Sohal RS . Relationship between mitochondrial Superoxide and hydrogen peroxide production and longevity of mammalian species. Free Radic Biol Med 1993;15:621–7.
Barja G, Cadenas S, Rojas C, Perez-Campo R, Lopez-Torres M . Low mitochondrial free radical production per unit O2 consumption can explain the simultaneous presence of high longevity and high aerobic metabolic rate in birds. Free Radic Res 1994;21:317–27.
Hart RW, Setlow RB . Correlation between deoxyribonucleic acid excision-repair and life-span in a number of mammalian species. Proc Natl Acad Sci USA 1974;71:2169–73.
Cortopassi GA, Wang E . There is substantial agreement among interspecies estimates of DNA repair activity. Mech Ageing Dev 1996;91:211–8.
De Murcia G, Shall S . Poly(ADP-ribosylation) reactions: from DNA damage and stress signalling to cell death. Oxford: Oxford University Press, 2000.
Bürkle A . Poly(ADP-ribosyl)ation: a posttranslational protein modification linked with genome protection and mammalian longevity. Biogerontology 2000;1:41–6.
Molinete M, Vermeiden W, Bürkle A, Ménissier-de Murcia J, Küpper J-H, Hoeijmakers JHJ, et al. The poly(ADP-ribose) polymerase DNA-binding domain blocks alkylation-induced DNA repair synthesis in living cells. EMBO J 1993;12:2109–17.
Grube K, Bürkle A . Poly(ADP-ribose) polymerase activity in mononuclear leukocytes of 13 mammalian species correlates with species-specifics life span. Proc Natl Acad Sci USA 1992;89:11759–63.
Beneke S, Alvarez-Gonzalez R, Bürkle A . Comparative kinetic characterisation of baculovirus-expressed human and rat poly(ADP-ribose) polymerase-1. Exp Gerontol 2000;35:989–1002.
Meyer R, Müller M, Beneke S, Küpper JH, Bürkle A . Negative regulation of alkylation-induced sister-chromatid exchanges by poly(ADP-ribose) polymerase-1 activity. Int J Cancer 2000;88:351–5.
Bürkle A . Poly(ADP-ribosyl)ation, genomic instability, and longevity. Ann N Y Acad Sci 2000;908:126–32.
Kirkwood TB . Molecular gerontology: bridging the simple and the complex. Ann N Y Acad Sci 2000;908:14–20.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bürkle, A. Mechanisms of ageing. Eye 15, 371–375 (2001). https://doi.org/10.1038/eye.2001.139
Issue Date:
DOI: https://doi.org/10.1038/eye.2001.139
This article is cited by
-
Editorial
Eye (2001)